Kristina Campbell, microbiome science writer@bykriscampbell
Sep 23, 2022
8 tweets
 A mini thread on the big news today in microbiome therapeutics...
To date, no live biotherapeutic products have been approved to go on the market as drugs.
But the FDA's Vaccines and Related Biological Products Advisory Committee held a meeting today... a LONG meeting.
A mini thread on the big news today in microbiome therapeutics...
To date, no live biotherapeutic products have been approved to go on the market as drugs.
But the FDA's Vaccines and Related Biological Products Advisory Committee held a meeting today... a LONG meeting.
 A mini thread on the big news today in microbiome therapeutics...
To date, no live biotherapeutic products have been approved to go on the market as drugs.
But the FDA's Vaccines and Related Biological Products Advisory Committee held a meeting today... a LONG meeting.
A mini thread on the big news today in microbiome therapeutics...
To date, no live biotherapeutic products have been approved to go on the market as drugs.
But the FDA's Vaccines and Related Biological Products Advisory Committee held a meeting today... a LONG meeting.At this meeting the committee members discussed the @Rebiotix Inc. / @Ferring Pharmaceuticals drug candidate called Rebyota -- a "standardized, stabilized" version of fecal microbiota transplantation -- to decide whether to recommend market approval.
The discussions centered around 2 questions...
1) Are the available data adequate to support the EFFECTIVENESS of Rebyota to reduce recurrence of C. difficile infection in adults following antibiotic treatment for recurrent CDI?
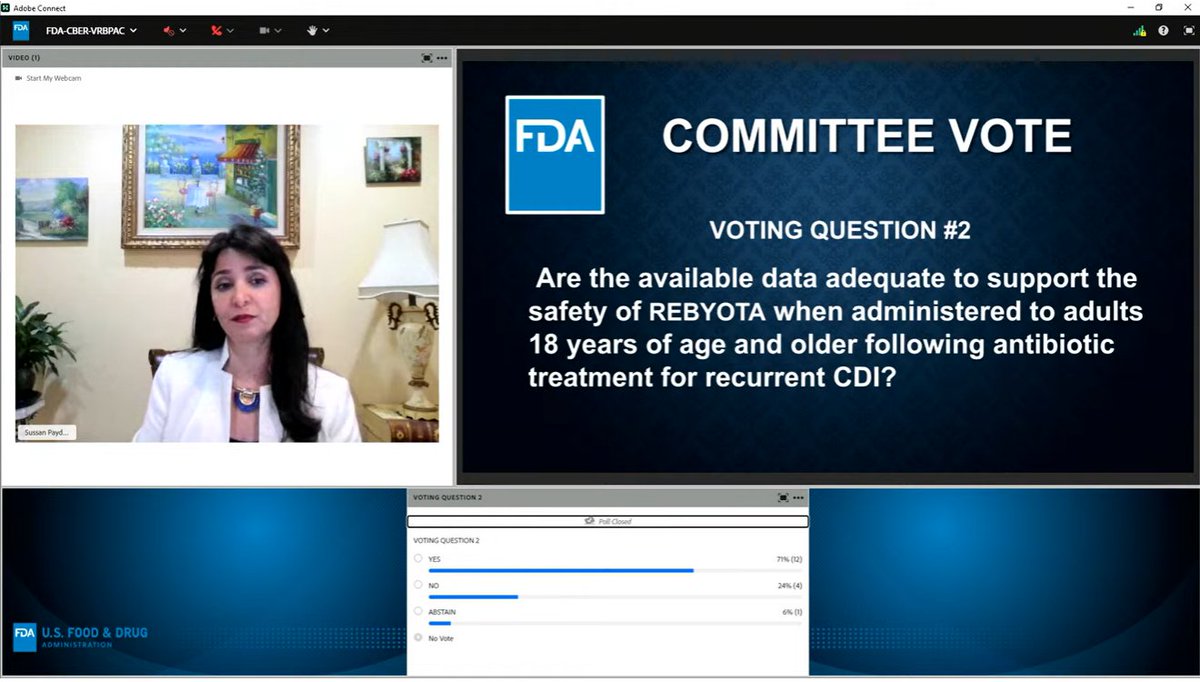
2) Are the available data adequate to support the SAFETY of Rebyota for this indication?
Committee members spent a long while discussing the data presented by the company for safety & efficacy. The data weren't convincing to all of the committee members -- they called the trials "imperfect".
But perhaps necessarily imperfect, given difficulties with recruitment, etc.
But after that...
the patients spoke. Several people shared their personal horror stories of recurrent C. difficile, including such anecdotes as:
"Chained to the toilet"
"Lost 3 years of my life"
"The toll it took on me and my family was enormous"


So it seemed committee members were faced with some okay data but a VERY clear message about the need for better treatment options.
Then came the vote. I was live-streaming the meeting, and I was literally on the edge of my seat with my heart pounding.
The efficacy question? Approved by 13 / 17 committee members.
The safety question? Approved by 12 / 17 committee members.
Approved.
This is a first. It's the closest a microbiome-focused therapy has ever come to the market... and reaching patients who cannot get, or won't get, or won't benefit from FMT.
There are more therapies following closely on this one.
Should be an exciting time ahead for this field 


Kristina Campbell, microbiome science writer
@bykriscampbell
Reporting on the front lines of microbiome science.
Health, diet, medicine, biotech.
Books: Well-Fed Microbiome & Gut Microbiota textbook for dietitians.
She.
Missing some tweets in this thread? Or failed to load images or videos? You can try to .